Partnerships in the Introduction of New Routine Vaccines in Bangladesh: Evidence from a Prospective Process Evaluation
International Centre for Diarrhoeal Disease Research, Bangladesh, or icddr,b (Luies, Uddin); Bangladesh Institute of Governance and Management (Sultana); D'EVA Consulting (Budden); iDE Bangladesh (Asaduzzaman); Shahjalal University of Science and Technology (Hossain); The Australian National University (Kelly, Gray, Sarma)
"Partners have played vital roles during country readiness...and successful launch of the vaccine[s]..."
With support from Gavi and its partners, the Government of Bangladesh (GOB) jointly introduced two vaccines in 2015: pneumococcal 10-valent conjugate vaccine (PCV-10) and inactivated polio vaccine (IPV). This study of the joint introduction of PCV-10 and IPV documents how Gavi's resources, processes, and partnership function in Bangladesh and shares lessons for strengthening vaccine introductions and routinisation in Bangladesh and elsewhere. Its focus is on immunisation partnership, which it defines as the contribution and engagement of the key development partners in delivering technical assistance as per the country's needs. The study is part of a prospective process and outcome evaluation conducted for the Gavi Full Country Evaluations (FCE) between 2013 and 2016.
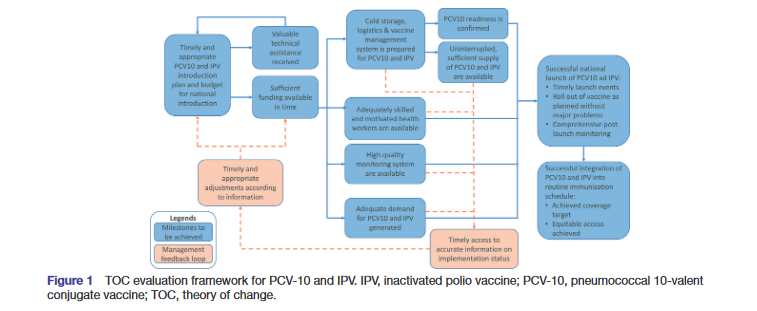
The researchers collaboratively developed a theory of change (TOC) framework of new vaccine introduction related to PCV-10 and IPV within the FCE consortium, depicting programme implementation milestones and indicators. The TOC framework defines the successful introduction of PCV-10 and IPV based on three criteria: (i) timely implementation of launch events such as the launch ceremony occurring as planned; (ii) achieving targets for the roll-out of the vaccine as planned without significant problems, such as lack of demand for the vaccine, and (iii) implementing comprehensive postlaunch monitoring activities, such as postlaunch supervision and a postintroduction evaluation.
The researchers triangulated and synthesised process tracking information, prioritised issues requiring deeper investigation, and used root cause analysis (RCA) to identify underlying causes of challenges and successes. They then conducted 19 key informant interviews (KIIs) and focus group discussions (FGDs) with 16 key stakeholders and service providers to validate and confirm assumptions and hypotheses from the RCA and develop recommendations for adaptive actions. They also reviewed relevant documents and observed trainers' and vaccinators' training and key stakeholder meetings. The study focused on eight Upazila (subdistrict) and one city corporation covering nine districts and seven administrative divisions of Bangladesh.
The study found that the Expanded Programme on Immunization (EPI) experienced several challenges during the joint introduction of PCV-10 and IPV, such as frequent changes in the vaccine introduction schedule, delays in budget allocation, vaccine supply shortage, and higher wastage rates of IPV. However, the timely response of EPI partners like the World Health Organization (WHO) and United Nations Children's Fund (UNICEF), drawing on their respective multicountry experiences, helped mitigate the challenges without generating additional risk to the programme.
For example, EPI stakeholders and partners organised joint advocacy meetings, including in urban municipalities and city corporations, within a short period, before the launching of vaccine. The advocacy meetings were conducted for mass social mobilisation. All health and family planning officials and workers participated in these sessions, including non-governmental organisation (NGO) representatives, journalists, respected personnel from Upazila administration, officers in-charge of police stations, municipality mayors, and religious leaders. The joint advocacy was possible because of strong commitment and timely support from UNICEF, the WHO, and other government ministries.
Based on the investigation, the researchers identify five key characteristics for a successful partnership: motivation for improvement, long-term commitment, financial and technical support, partners' mandate to support the government, and coordination. In Bangladesh, the WHO and UNICEF are long-term partners of EPI and have, over the years, built commitment and trust. They maintain strong coordination with the relevant ministries and local government bodies and other departments, which helps them to better communicate any specific situation and need-based adjustments. All partners were on board and actively involved in discussions, decision-making, and policy changes regarding any new vaccine introductions.
In conclusion: "The partnership among the EPI stakeholders played an essential role in the joint introduction of PCV-10 and IPV, besides strengthening the immunisation systems in Bangladesh. Despite some challenges in implementation, the vaccine roll-out proceeds according to the TOC milestones. Development partners act rapidly with their technical assistance throughout and they expedite the implementation process by developing the training and communication materials....With all these essential activities and supports, Bangladesh has been able to successfully introduce two new vaccines at the first time. These accomplishments are attributed to various factors that should be supported in the upcoming Gavi-supported introductions of new vaccines in Bangladesh. The lessons learnt can be used in other countries...introducing new vaccines."
BMJ Open 2022;12:e061742. doi:10.1136/bmjopen-2022-061742.
- Log in to post comments












































